Cholesterol-Conjugated sdRNA
Modified double-stranded RNA conjugated with cholesterol for enhanced cellular uptake without the need for viral vectors or lipid nanoparticles.
Targeted Gene Silencing
Specifically targets genes that modulate scarring and fibrosis, promoting regenerative healing while minimizing off-target effects.
Extended Duration
Chemical modifications enable pharmaceutical benefit lasting >90 days in vivo, dramatically reducing dosing frequency.
Explore how our sdRNA technology works, from administration to therapeutic effect

sdRNA Design & Synthesis
Our proprietary self-delivering RNA (sdRNA) molecules are designed to target an enzyme controlling multiple proteins involved in the fibrotic pathway. A hydrophobic conjugation to the sdRNA backbone optimizes cellular uptake without the need for AAV or LNP delivery technology.
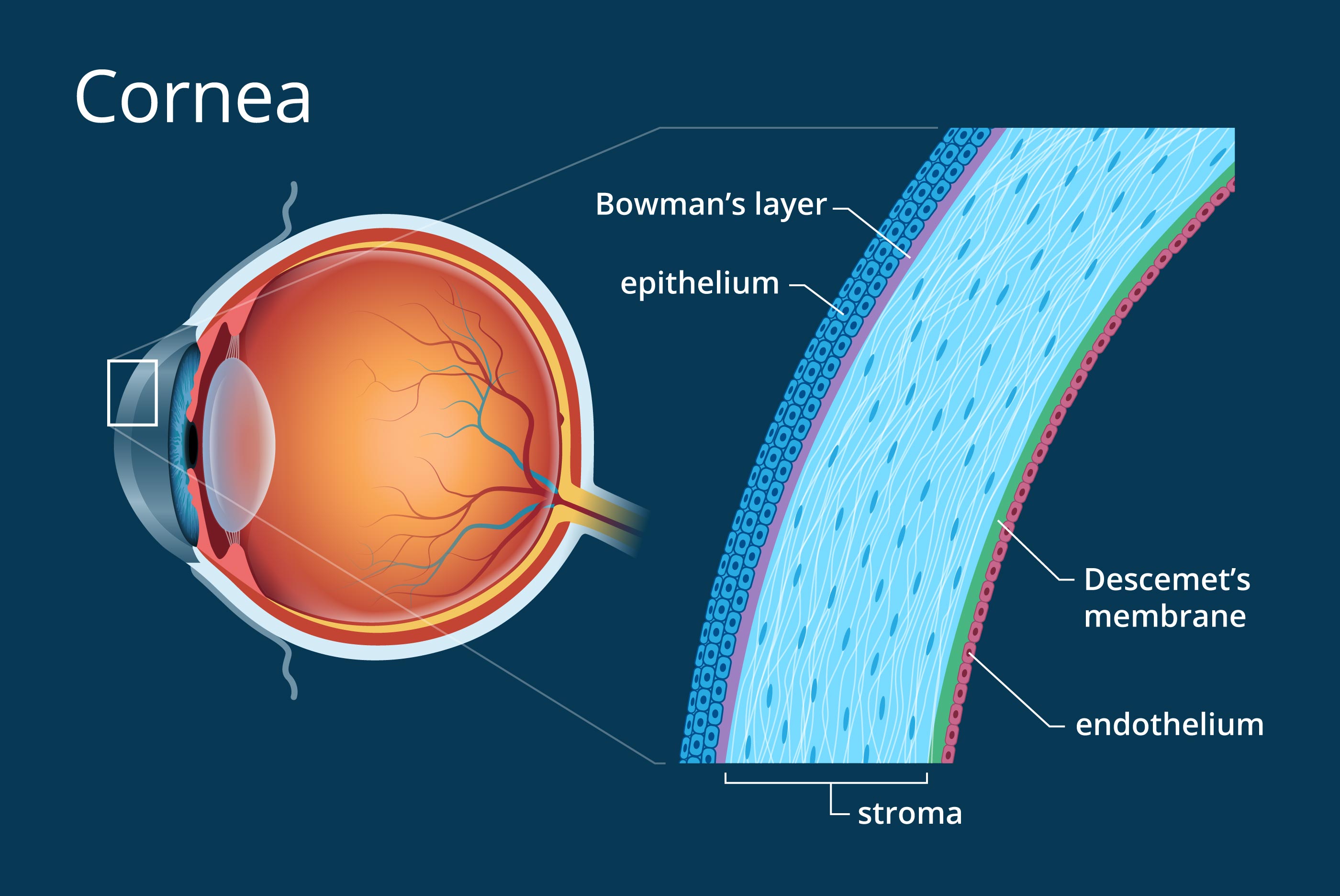
Cornea Anatomy: Our sdRNA targets the stroma layer where scarring occurs
Understanding Corneal Structure
The cornea is a complex, multi-layered tissue that must remain transparent for clear vision. When injured, the stroma—the thickest layer—can develop scar tissue that clouds vision permanently.
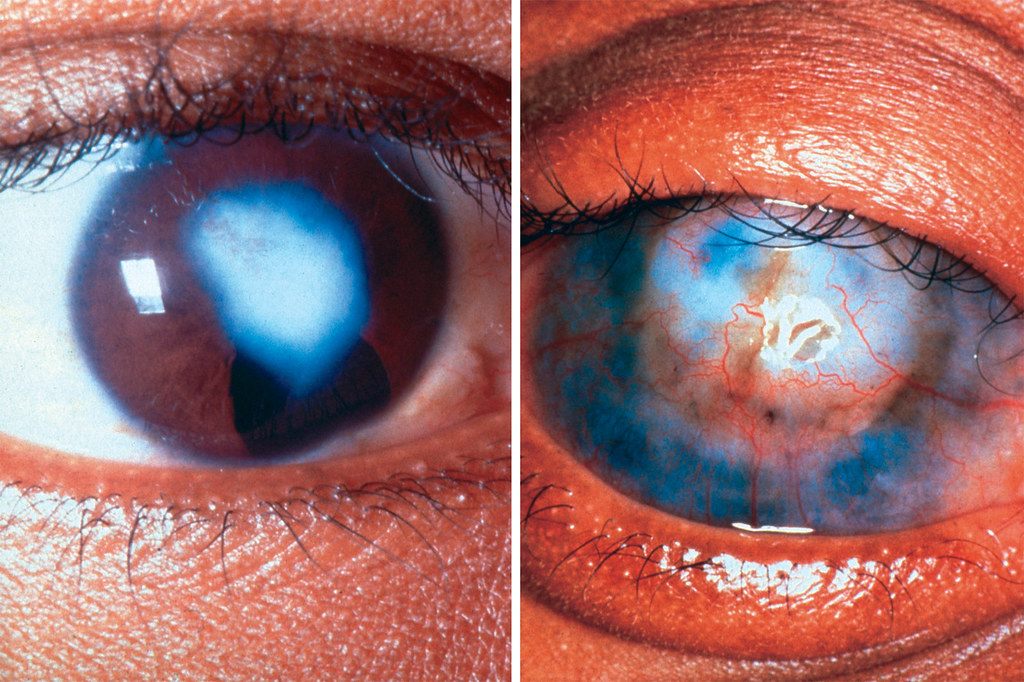
Healthy vs. Scarred Cornea
Corneal scarring results from abnormal wound healing, where excessive collagen deposition creates opaque tissue. Our sdRNA technology intervenes at the genetic level to promote regenerative healing instead of scarring.
- Traditional treatments only manage symptoms
- Corneal transplants have limited availability
- DUB's sdRNA prevents scarring at the source

Precise Targeting
Designed to target specific genes involved in fibrosis and scarring
Self-Delivering
No viral vectors or lipid nanoparticles needed—cholesterol conjugation enables direct cellular uptake
Potent Silencing
Achieves significant gene knockdown through RNA interference pathway
Long-Lasting
Single treatment provides therapeutic benefit for over 90 days
| Feature | DUB Tx | Steroids | NGF Therapy | Transplant |
|---|---|---|---|---|
| Topical Delivery | ||||
| Effective Treatment | Mixed | |||
| Single Dose | ||||
| Affordable | ||||
| Room Temperature Stable | ||||
| Reduces Scarring | Partial | |||
| Accessible |
Research & Development
Backed by rigorous scientific research and NIH funding
Preclinical Studies
Extensive preclinical validation demonstrating efficacy in corneal wound healing models. Studies show significant reduction in scarring and improved healing rates compared to standard of care.
- Demonstrated >90-day pharmaceutical benefit in vivo
- No signs of toxicity in current experiments
- Room temperature stability validated
NIH-Funded Research
$2 million NIH National Eye Institute grant supporting investigation of sdRNA for glaucoma surgery scarring prevention. This research expands our platform's applicability to additional high-need indications.
Clinical Development
Target clinical trial initiation in 2027 for persistent corneal epithelial defect (PCED) indication. Regulatory strategy developed with experienced FDA consultants.

Precision Medicine: Targeting the cornea at the molecular level
Key Research Milestones
- Proof of concept validated
- Lead candidate identified
- IND-enabling studies in progress